-
甘蔗(Saccharum officinarum L.)是世界上最重要的一类糖料经济作物。全世界75%的糖来源于甘蔗,中国糖业对国内生产总值(GDP)的贡献约为60~80亿元人民币,约占中国国内生产总值(GDP)的0.1%[1]。甘蔗虫害严重危害甘蔗产量,影响甘蔗品质,随着转基因技术的日益成熟,利用转基因的方法将外源抗虫基因导入甘蔗基因组,能够有效的防治甘蔗虫害,提高甘蔗产量。然而,转基因作物带来的生态安全问题不容小觑,这其中就包括转基因作物对土壤生态系统带来的影响,主要表现为对土壤的养分、理化性质、酶活性、土壤中动植物及微生物的影响。近年来,转基因作物对土壤生态系统的影响已成为对转基因作物进行安全性评价的研究热点[2]。土壤微生物参与有机质分解、腐殖质形成、养分转化和循环等一系列生化过程,是土壤生物活性的重要组成部分。土壤酶主要由土壤微生物及植物根系产生,土壤酶活性与土壤其他性质密切相关,如容重、土壤pH值、养分和SOM的分布等[3-4]。土壤酶活性反映了土壤内有机质的动态变化以及土壤内C,N,S,P等元素的物质循环,是评价土壤质量的重要指标。根际是植物、土壤和微生物相互作用的关键区域。外源基因可能通过根系分泌物、植物掉落的残体或花粉进入土壤[5-7]。研究转基因作物根际土壤酶活性,有助于了解外源表达基因产物对土壤微生态的影响。目前,已有许多研究报道了转Bt基因作物对土壤酶活性的影响,但实验结果因不同的转基因作物甚至不同的转基因株系而存在很大的差异。就Bt转基因玉米而言,颜世磊等[8]在大田自然条件下比较研究了转Bt基因玉米种植和秸秆分解时对土壤酶活性影响的差异。结果表明,在喇叭口期和抽雄期,土壤蔗糖酶和土壤酸性磷酸酶活性显著提高。在秸秆还田后,土壤蔗糖酶的活性则有显著提高。作者认为Bt玉米及秸秆还田对土壤酶活性存在影响,影响的幅度及趋势随玉米生育期和土壤酶种类的不同而产生差异,并指出商业化Bt玉米的环境释放仍有待长期定位观测和评价;范巧兰等[9]也认为转Bt基因玉米对土壤酶活性有一定的影响。推测种植Bt玉米对土壤酶活性的影响可能是因根系分泌的Bt蛋白引起土壤微生物多样性的变化而导致,另一种推测是外源 Bt基因导入后引起玉米秸秆以及根系分泌物化学成分的差异而造成;刘玲[10]等认为转Bt玉米对碱性磷酸酶在个别生育期影响显著。然而Flores等[11]、Icoz等[12]则认为转基因玉米对土壤酶活性没有显著影响。而Bt转基因棉花对根际土壤酶活性的影响同样存在不一致的现象。范巧兰等[2]、万小羽等[13]、Sun等[7]认为转基因棉花不同生育期对土壤酶活性有一定的影响;Shen等[14]则认为转基因棉花对土壤脲酶、碱性磷酸酶、蛋白酶等活性没有影响。转Bt基因甘蔗的研究跟其他作物相比相对落后,仅见Zhou等[15] 报道了关于转基因cry1Ac抗虫甘蔗对土壤脲酶、蛋白酶、蔗糖酶活性等的影响,实验结果显示与对照比较,不同的转基因甘蔗株系在个别生长时期的根际土壤酶活性存在显著差异,但总体而言多数土壤样品中没有显着差异。众所周知,甘蔗是多年宿根作物,因此转Bt基因甘蔗一般也会宿根种植3~4年,这种情况下极有可能导致Bt蛋白在土壤中富集,从而影响土壤的生物活性。因此为了更确切地探究种植转Bt基因甘蔗对根际土壤酶活性的影响,本研究以种植转Cry1Ac-2A-gna基因甘蔗优良株系Bt2,Bt17连续宿根3 a的大田土壤作为研究对象,测定不同生长时期转Cry1Ac-2A-gna基因甘蔗对其根际土壤酶活性的影响,为株系Bt2、Bt17的环境释放提供科学的数据支持。
-
海南省文昌市迈号镇乌鸡塘村农业部转基因植物及植物用微生物环境安全监督检验测试中心(海口)试验基地。试验基地土壤类型为冲积土,土壤质地为沙壤土,土壤有机质含量较低,为(0.82±0.1)%、pH为6.5±0.1,含水量为(12±0.01)%。地处东经108°21′至111°03′,北纬19°20′至20°10′之间,属热带季风岛屿型气候,光、水、湿、热条件优越,全年无霜冻,四季分明。年平均温度23.9 ℃,最低极温0.3~6.6 ℃,出现在1月份。年平均日照1 953.8 h。夏日日照最长是13.19 h,冬日日照仅10.57 h。年太阳辐射总能量为455.4~481.3 kJ·cm−2。雨量丰富,但时空分布不均,干、湿季明显,春旱突出,常年降雨量1 721.6 mm,平均1 529.8~1 948.6 mm。
-
转Cry1Ac-2A-gna融合基因甘蔗Bt2,Bt17号株系T3代植株,对照为受体植株甘蔗新台糖ROC22。
-
抗虫转基因甘蔗优良株系的种植采用随机区组设计,3次重复,四周设保护行。每个小区种植转基因株系4行,行长10 m,行距为1.2 m,该转基因甘蔗在试验基地已宿根连续3 a种植。在T3代甘蔗不同的生育期,即苗期、分蘖期、生长期、成熟期采取根际深度为10~15 cm的土壤,每个样品采3丛甘蔗的根际土壤混合,每个株系3个重复,带回实验室自然风干后,过筛,储存于4 ℃,用于测定土壤酶活性,非转基因甘蔗根际土壤作为对照。
-
测定抗虫转基因甘蔗根际土壤蔗糖酶、酸性磷酸酶、中性磷酸酶、碱性磷酸酶活性,分别采用索莱宝公司的土壤蔗糖酶(S-SC)活性检测试剂盒、土壤酸性磷酸酶(S-ACP)活性检测试剂盒、土壤中性磷酸酶(S-NP)活性检测试剂盒及土壤碱性磷酸酶(S-AKP/ALP)活性检测试剂盒进行测定。
-
采用Excle2007及SPSS22.0统计分析测定数据。
-
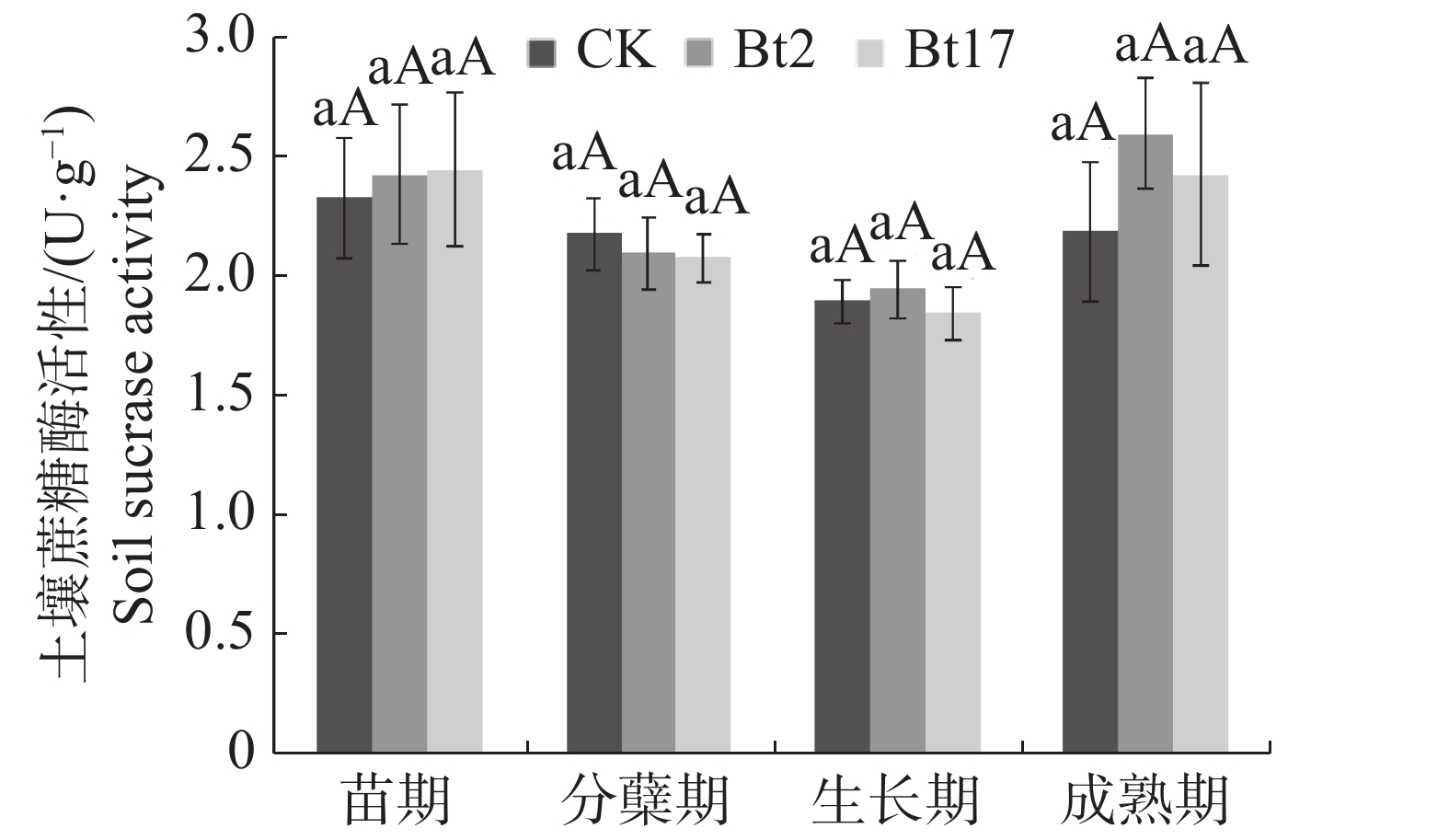
土壤蔗糖酶能够促进土壤中的蔗糖转化为葡萄糖和果糖。土壤的呼吸强度会随着土壤中N,P元素的增加,土壤微生物数量的增加或土壤中有机质的增加而增加,同时蔗糖转化为葡萄糖及果糖的效率也越高[16]。土壤蔗糖酶活性在一定程度上反映了土壤中有机质的代谢强弱,与土壤中的C素循环相关,间接反映土壤肥力及土壤的熟化程度。试验结果表明,抗虫转基因甘蔗优良株系Bt2及Bt17在甘蔗生长的苗期、分蘖期、生长期及成熟期的土壤蔗糖酶活性与受体甘蔗ROC22相比并无差异(图1)。在甘蔗整个生育期抗虫转基因甘蔗Bt2与Bt17的土壤蔗糖酶活性呈现在苗期至生长期下降,生长期至成熟期上升的趋势。土壤蔗糖酶活性在Bt2,Bt17两个株系间也没有差异,说明外源抗虫基因Cry1Ac及gna的导入并不会对根际土壤的蔗糖酶活性产生影响。
-
土壤磷酸酶与土壤中有机磷的转化密切相关,能加速有机磷的脱磷速度[17],土壤磷酸酶活性受到土壤pH值以及土壤中有机磷的含量影响,与土壤中磷素循环相关,反映了磷素的生物转化方向及强度[18-20]。土壤磷酸酶的最适pH范围有3种,分别为pH4~5,pH6~7和pH8~10。土壤磷酸酶最大活性的测定要在相应pH缓冲液条件下,分别测定土壤酸性、中性、碱性磷酸酶的活性,才能测出土壤磷酸酶的最大活性。
-
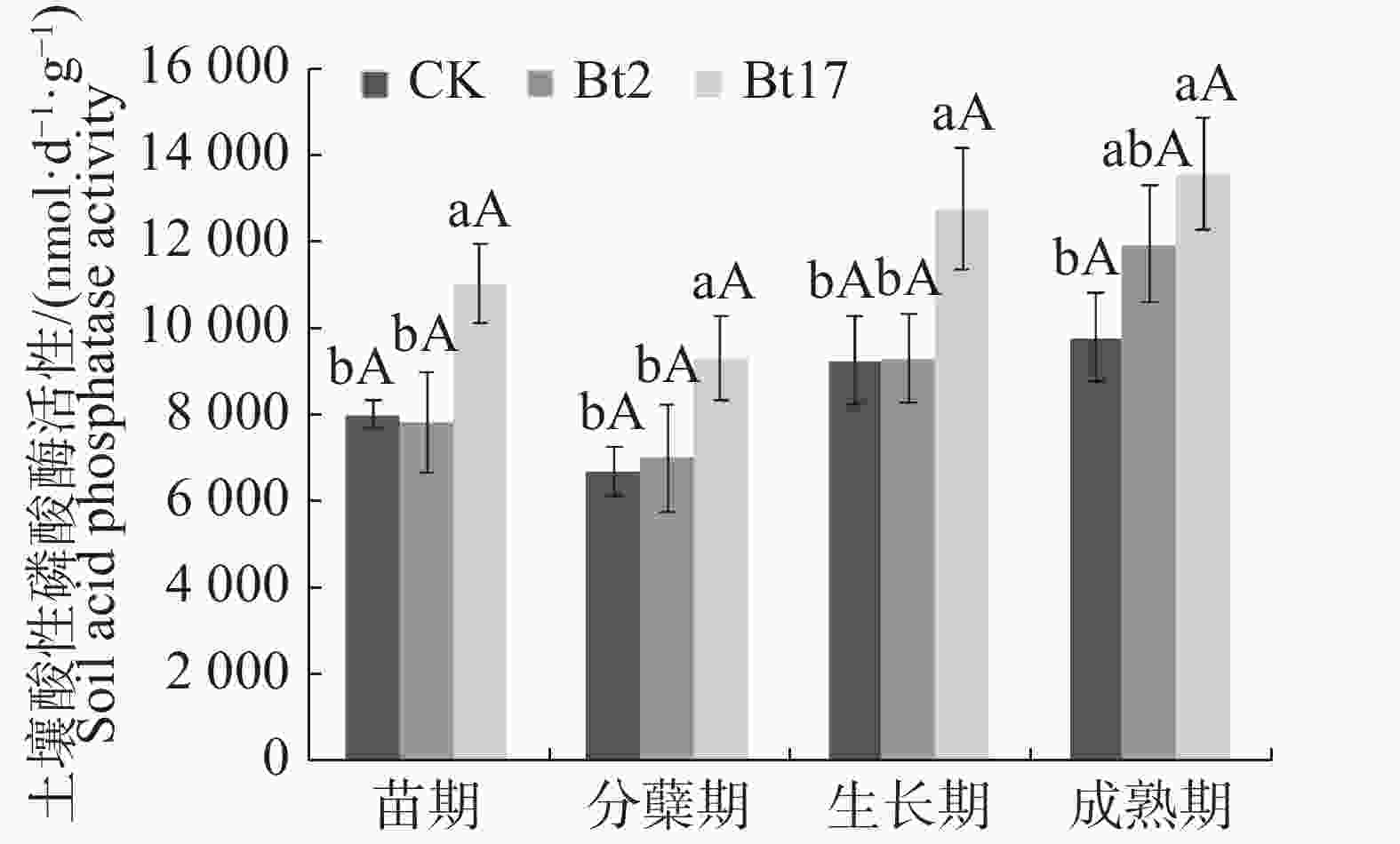
由图2可见,苗期时,抗虫转基因甘蔗Bt2土壤酸性磷酸酶活性与非转基因对照并无差异,Bt17则显著高于对照37.50%,并且Bt17显著高出Bt2 40.52%;分蘖期时,Bt2土壤酸性磷酸酶与对照无差异,Bt17则显著高出对照38.11%,显著高出Bt2 32.54%;生长期时,Bt2土壤酸性磷酸酶活性与对照并无差异,Bt17则显著高于对照33.86%,并且Bt17显著高出Bt2 36.97%。成熟期时,Bt2土壤酸性磷酸酶与对照无差异,Bt17显著高出对照39.00%,与Bt2相比无差异。实验结果表明,抗虫转基因甘蔗Bt2号株系不会对土壤酸性磷酸酶活性产生影响,而Bt17在甘蔗生长的各个时期的土壤酸性磷酸酶活性均显著高于对照;2个抗虫转基因甘蔗株系(Bt2,Bt17)的土壤酸性磷酸酶在甘蔗整个生育期呈现相同的变化趋势,2株系的土壤酸性磷酸酶活性均在苗期至分蘖期时呈现下降的变化趋势,在分蘖期至成熟期呈现上升的变化趋势。
-
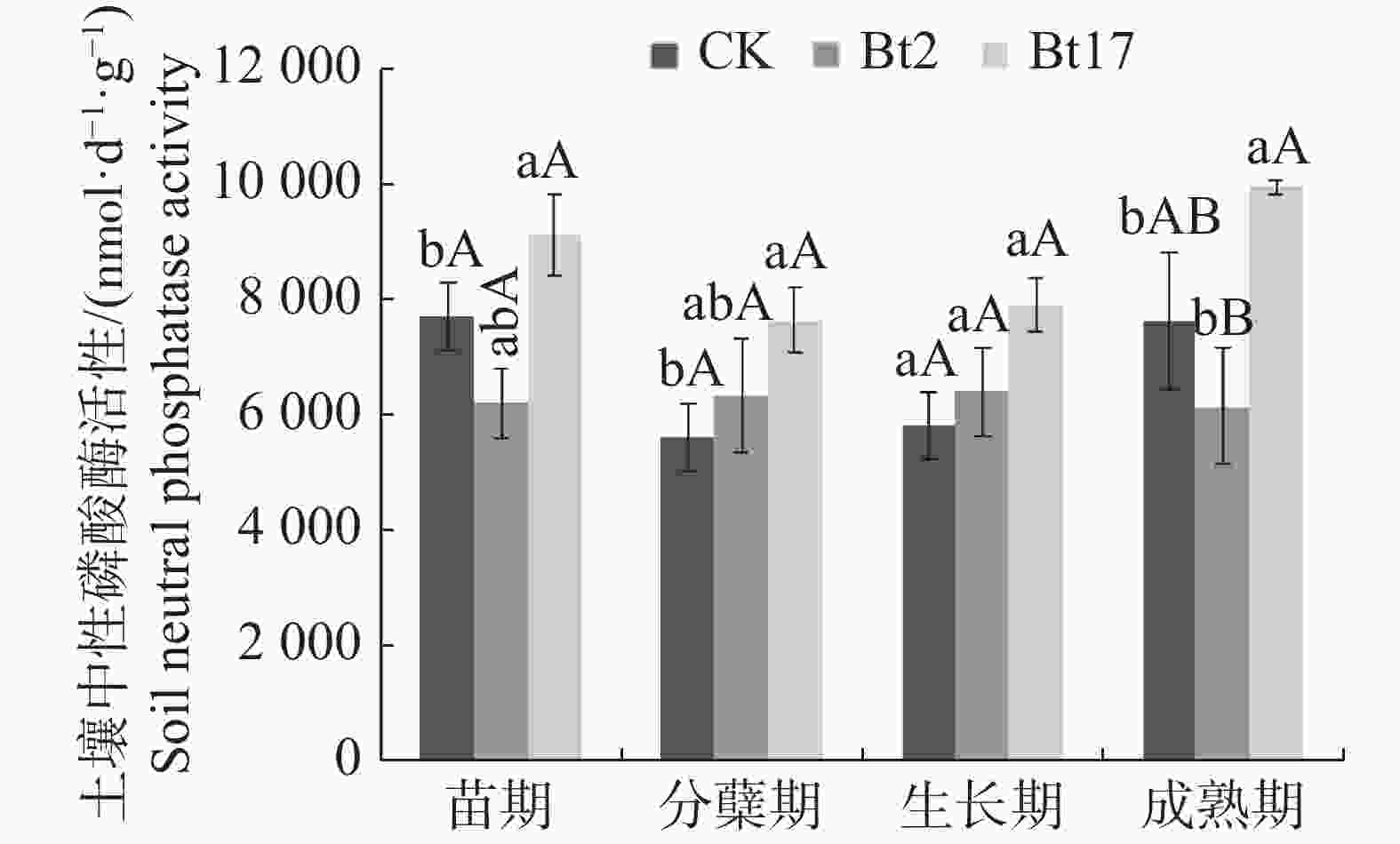
由图3可见,苗期时,抗虫转基因甘蔗Bt2的土壤中性磷酸酶活性与非转基因对照无差异,抗虫转基因甘蔗Bt17则显著高于对照18.11%,但Bt17与Bt2的土壤中性磷酸酶活性并无差异;分蘖期时,抗虫转基因甘蔗Bt2的土壤中性磷酸酶与对照无差异,抗虫转基因甘蔗Bt17则显著高出对照35.77%,Bt17与Bt2的土壤中性磷酸酶活性并无差异;生长期时,抗虫转基因甘蔗Bt2、Bt17的土壤中性磷酸酶活性与对照相比均无差异;成熟期时,抗虫转基因甘蔗Bt2的土壤中性磷酸酶的活性与对照无差异,抗虫转基因甘蔗Bt17的土壤中性磷酸酶的活性则显著高出对照30.36%,且极显著高出Bt2 61.42%。实验结果表明,抗虫转基因甘蔗Bt2不会对土壤中性磷酸酶活性产生影响,而抗虫转基因甘蔗Bt17的土壤中性磷酸酶活性在甘蔗的苗期、分蘖期、成熟期均显著高于对照,但在甘蔗的生长期时与对照无差异;2个抗虫转基因甘蔗株系土壤中性磷酸酶在甘蔗的整个生育期呈现不同的变化趋势。Bt2在甘蔗整个生长时期,其土壤中性磷酸酶活性变化基本稳定不变,而Bt17土壤中性磷酸酶活性在苗期至分蘖期时呈现下降的趋势,在甘蔗分蘖期至成熟期时整体呈现土壤中性磷酸酶活性上升的趋势。
-
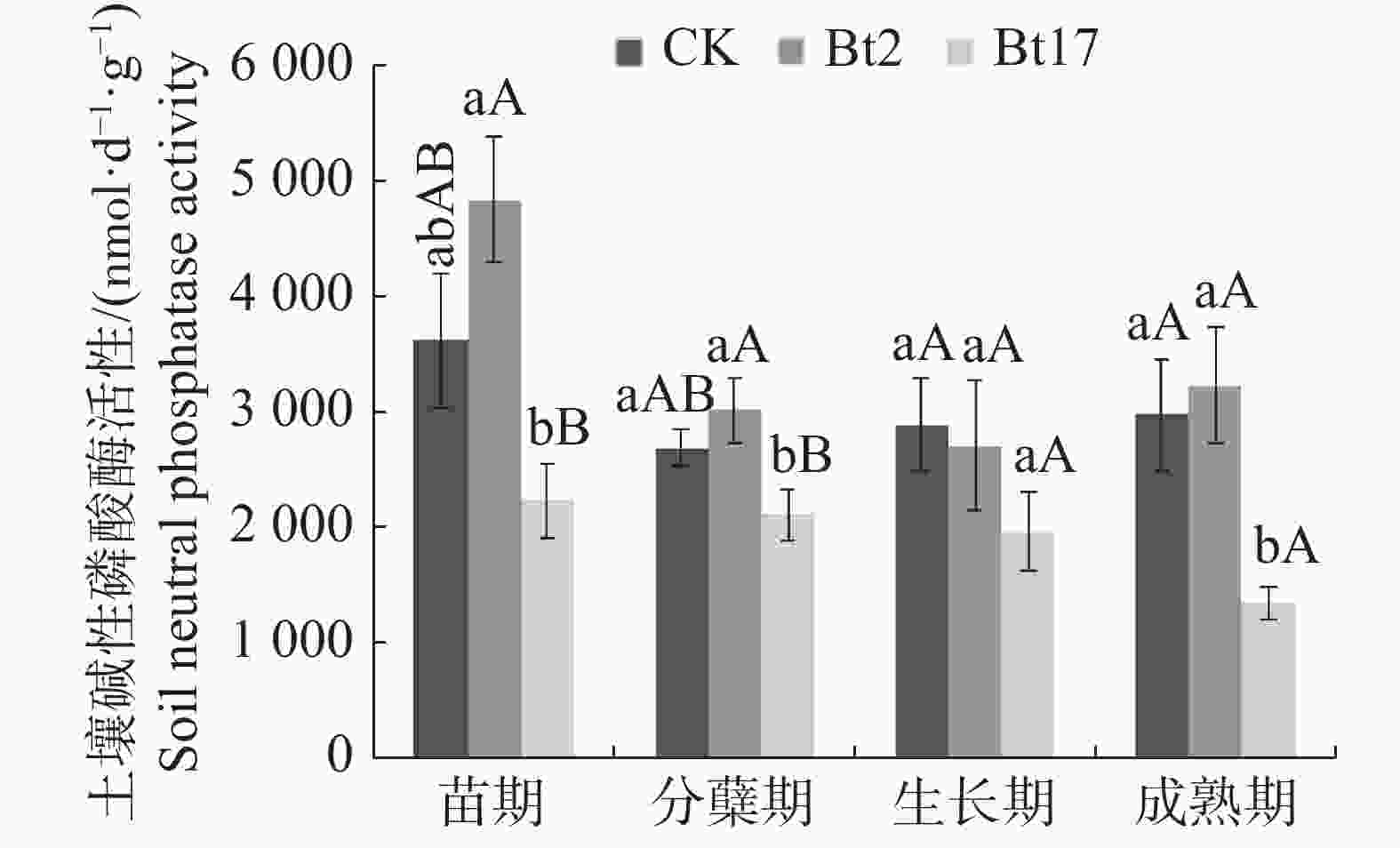
由图4可见,苗期时,抗虫转基因甘蔗Bt2、Bt17的土壤碱性磷酸酶活性与非转基因对照无显著性差异,但Bt17极显著低于Bt2 53.68%;分蘖期时,Bt2土壤碱性磷酸酶与对照无显著性差异,Bt17则显著低于对照21.44%,极显著低于Bt2 30.02%;生长期时,Bt2,Bt17的土壤碱性磷酸酶活性与对照相比均无显著性差异;成熟期时,Bt2的土壤碱性磷酸酶与对照无显著性差异,但Bt17显著低于对照54.49%,并且显著低于Bt2 58.13%。实验结果表明,抗虫转基因甘蔗Bt2不会对土壤碱性磷酸酶活性产生影响,而Bt17在甘蔗生长的分蘖期、成熟期其土壤碱性磷酸酶活性均显著低于对照,苗期与生长期时不会对土壤碱性磷酸酶活性产生影响;2个抗虫转基因甘蔗株系土壤碱性磷酸酶在甘蔗的整个生育期呈现不同的变化趋势,Bt2在甘蔗生长的苗期至生长期土壤碱性磷酸酶活性呈现下降的趋势,生长期至成熟期呈上升趋势;Bt17土壤碱性磷酸酶活性则在甘蔗的整个生长周期呈现逐渐平缓下降的趋势。
-
土壤酶通常是转基因植物的风险评估的一项重要内容,因为它们参与土壤养分循环。土壤是土壤中的生物与外界环境因子发生物质循环及能量转化的场所。转基因作物的外源基因是否会通过转基因作物根系分泌物、作物残渣等转移到土壤中并在土壤中积累,这可能会对土壤环境产生一定的影响[21]。它可能会通过影响土壤酶活性,影响土壤肥力、改变土壤的生物功能类群或改变土壤生物多样性,另一方面转基因植物和非转基因植物在整个发育过程中的根际土壤酶活性可能会受土壤化学性质差异的影响,如pH,粘土矿物组成,或其他一些自然因素的影响,如季节性变化,降雨量和分布[22]。阮妙鸿等[23]对转甘蔗花叶病毒外壳蛋白基因(ScMV-CP)的转基因甘蔗进行了对根际土壤酶活性影响的测定,认为转基因甘蔗显著降低了土壤脱氢酶的活性,提高了土壤纤维素酶、蛋白酶和脲酶的活性,而对土壤磷酸单脂酶和蔗糖酶没有明显影响,总体而言转基因甘蔗对根际土壤的酶活性有一定的影响。由于该实验仅对转ScMV-CP基因甘蔗的生长期进行测定,因此该实验结果并未能全面体现转ScMV-CP基因甘蔗的整个生育期各个时期对根际土壤酶活性影响;Zhou等[15]首次报道了关于转基因cry1Ac抗虫甘蔗对土壤脲酶、蛋白酶、蔗糖酶活性等的影响,与对照相比,多数转基因株系的根际土壤酶活没有明显的差异。但在一些株系的某些生长阶段存在显著性差异,具体表现为转基因甘蔗品系与非转基因甘蔗在整个发育过程中根际土壤酶活性的变化规律没有一致的趋势;同一转基因品系的活性随着发育阶段的变化而变化或同一阶段的土壤酶活性随着转基因甘蔗品系的变化而变化。Zhou等[15]推测土壤酶活性的变化可能是由于土壤化学性质如pH,肥料添加或和其他一些自然因素的差异造成的,认为转BT基因甘蔗对土壤酶的负面影响很小。
笔者对转Cry1Ac-2A-gna基因的2个甘蔗株系的整个生育期对根际土壤的酶活性进行跟踪测定。与对照相比,抗虫转基因甘蔗株系Bt17的根际土壤酶活在不同的生育期中存在显著性差异,具体表现在Bt17株系的土壤蔗糖酶在甘蔗生长的各个时期与对照均不存在显著性差异;土壤酸性磷酸酶活性和中性磷酸酶活性在多个生长期显著高出对照;土壤碱性磷酸酶活性在分蘖期和成熟期则显著低于对照。这说明Bt17对土壤酸性磷酸酶、中性磷酸酶和碱性磷酸酶可能有一定的影响。Bt2株系的土壤蔗糖酶、酸性磷酸酶、中性磷酸酶和碱性磷酸酶在甘蔗生长的各个时期与对照均不存在显著性差异,说明该转基因株系对上述的4种酶无影响。对于转基因甘蔗2个不同株系对土壤酶活性的影响不一致的研究结果,笔者推断可能是由于外源基因在根中的表达量差异而引起的,株系Bt17的cry1Ac在根中的表达量是株系Bt2的1.6倍,基因cry1Ac在株系Bt17的根中大量表达可能导致根系分泌物化学成分产生差异从而影响其根际土壤酶的活性;也有可能是由于土壤化学性质如pH,肥料添加或其他一些自然因素差异造成的。
分析转基因作物根际土壤酶活性一般选择测定蔗糖酶、磷酸酶、脲酶、蛋白酶、磷酸单酯酶、转化酶、过氧化氢酶、多酚氧化酶等酶活性。本实验目前已分析了蔗糖酶和3种磷酸酶,其余酶活性的测定仍然在进行中,通过后续工作的分析将会更全面地了解转基因抗虫甘蔗对根际土壤酶活性的影响。甘蔗一般宿根种植多年,转基因甘蔗是否对土壤酶活性、微生物群落结构等产生显著性影响目前尚无定论,因此开展转基因甘蔗对土壤生态系统的研究,获得较全面的转基因甘蔗安全性的基础数据,对转基因甘蔗的生产应用具有指导作用。
Effect of Insect-resistant Transgenic Sugarcane on Soil Enzyme Activities in the Rhizosphere
-
摘要: 为了评价抗虫转基因甘蔗优良株系Bt2、Bt17对土壤生态环境造成的生态安全风险,采集抗虫转基因甘蔗Bt2、Bt17号株系及其受体非转基因品种ROC22甘蔗根际附近土壤,研究抗虫转基因甘蔗对其根际土壤蔗糖酶、酸性磷酸酶、中性磷酸酶及碱性磷酸酶的影响。结果表明,抗虫转基因甘蔗对其根际土壤酶活性的影响会因甘蔗生长周期、抗虫转基因甘蔗株系以及酶的种类而大有不同。Bt2的土壤酶活性在甘蔗生长的各个时期均未与对照存在差异,而Bt17对土壤酶活性的影响则较为复杂。与受体非转基因甘蔗品种ROC22相比,Bt17根际的土壤蔗糖酶活性在甘蔗生长的任何时期都无差异;而土壤酸性磷酸酶活性在甘蔗生长的各个时期均显著高于对照;中性磷酸酶活性则在苗期、分蘖期、成熟期显著高出对照,而在生长期与对照无差异;土壤碱性磷酸酶活性在苗期、生长期时与对照无差异,但分蘖期和成熟期显著低于对照,说明Bt2号株系对土壤酶活性并未产生影响,Bt17号株系对土壤酶活性可能产生较小的影响。Abstract: In order to assess the ecological risks of insect-resistant transgenic sugarcane lines Bt2 and Bt17 on soil ecological environment rhizosphere soil samples were collected from plantations of insect-resistant transgenic sugarcane lines Bt2 and Bt17 and their non-transgenic donor sugarcane variety ROC22 to analyze the effect of the insect-resistant transgenic sugarcane lines on the activities of soil sucrase, acid phosphatase, neutral phosphatase and alkaline phosphatase in the rhizosphere. The results showed that the effects of the insect-resistant transgenic sugarcane lines on the soil enzyme activities in the rhizosphere varied greatly with the growth cycle of sugarcane, the insect-resistant transgenic sugarcane lines and the enzymes. Soil enzyme activity of sugarcane line Bt2 was not different from that of the control at all the stages of sugarcane growth, while the effect of Bt17 on soil enzyme activity was more complex. Compared with the non-transgenic donor sugarcane ROC22, the sugarcane line Bt17 was not different in soil sucrase activity in the rhizosphere at any stage of sugarcane growth, significantly higher in soil acid phosphatase activity at any stage of sugarcane growth, significantly higher in neutral phosphatase activity at seedling stage, at tillering stage and at mature stage, but not different in neutral phosphatase activity at the grand growth stage, but significantly lower in neutral phosphatase at the tillering stage and the mature stage. This indicated that the sugarcase line Bt2 had no effect on soil enzyme activity in the rhizosphere, while the sugarcane line Bt17 might have less effect on soil enzyme activity in the rhizosphere.
-
Key words:
- insect-resistant transgene /
- sugarcane /
- soil enzyme activity /
- ecological risk
-
-
[1] ZHANG M Q, GOVINDARAJU M. Sugarcane production in China[M/OL]//Alexandre Bosco de Oliveira. Sugarcane-Technology and Research, DOI: 10.5772/intechopen. 73113. (2018−05−16)[2019−04−15]. https://www.intechopen.com/books/sugarcane-technology-and-research/sugarcane-production-in-china [2] 范巧兰, 陈耕, 李永山, 等. 转Bt基因棉花对土壤酶活性的影响[J]. 农学学报, 2013, 3(1): 32 − 35. doi: 10.3969/j.issn.1007-7774.2013.01.007 [3] ACOSTA-MARTINEZ V, KLOSE S, ZOBECK T M. Enzyme activities in semiarid soils under conservation reserve program, native rangeland, and cropland [J]. Soil Biol. Biochem, 2003, 166: 699 − 707. [4] LEHMAN R M, CAMBARDELLA C A, STOTT, et al. Understanding and enhancing soil biological health: the solution for reversing soil degradation [J]. Sustainability, 2015, 7: 988 − 1027. doi: 10.3390/su7010988 [5] BLACKWOOD C B, BUYER J S. Soil microbial communities associated with Bt and non-Bt corn in three soils [J]. Journal of Environmental Quality, 2004, 33: 832 − 836. doi: 10.2134/jeq2004.0832 [6] GRIFFITHS B S, CAUL S, THOMPSON J, et al. Microbial and microfaunal community structure in cropping systems with genetically modified plants [J]. Pedobiologia, 2007, 51: 195 − 206. doi: 10.1016/j.pedobi.2007.04.002 [7] SUN C X, CHEN L J, WU Z J, et al. Soil persistence of Bacillus thuringiensis (Bt) toxin from transgenic Bt cotton tissues and its effect on soil enzyme activities [J]. Biology and Fertility of Soils, 2007, 43: 617 − 620. doi: 10.1007/s00374-006-0158-6 [8] 颜世磊, 赵蕾, 孙红炜, 等. 大田环境下转Bt基因玉米对土壤酶活性的影响[J]. 生态学报, 2011, 31(15): 4244 − 4250. [9] 范巧兰, 王慧, 李永山, 等. 转基因玉米对土壤酶活性的影响[J]. 山西农业科学, 2019, 47(1): 69 − 71. doi: 10.3969/j.issn.1002-2481.2019.01.17 [10] 刘玲, 赵建宁, 李刚, 等. 转Bt玉米对土壤酶活性及速效养分的影响[J]. 土壤, 2012, 44(1): 167 − 171. doi: 10.3969/j.issn.0253-9829.2012.01.028 [11] FLORES S, SAXENA D, STOTZKY G, et al. Transgenic Bt plants decompose less in soil than non-Bt plants [J]. Soil Biology & Biochemistry, 2005, 37: 1073 − 1082. [12] ICOZ I, SAXENA D, ANDOW D, et al. Microbial populations and enzyme activities in soil in situ under transgenic corn expressing Cry proteins fromBacillus thuringiensis [J]. Journal of Environmental Quality, 2008, 37: 647 − 662. doi: 10.2134/jeq2007.0352 [13] 万小羽, 梁永超, 李忠佩, 等. 种植转Bt基因抗虫棉对土壤生物学活性的影响[J]. 生态学报, 2007, 27(12): 5414 − 5420. doi: 10.3321/j.issn:1000-0933.2007.12.055 [14] SHEN R F, CAI H, GONG W H. Transgenic Bt cotton has no apparent effect on enzymatic activities or functional diversity of microbial communities in rhizosphere soil [J]. Plant and Soil, 2006, 285(1/2): 149 − 159. [15] ZHOU D, XU L, GAO S, et al. Cry1Ac transgenic sugarcane does not affect the diversity of microbial communities and has no significant effect on enzyme activities in rhizosphere soil within one crop season [J]. Frontiers in Plant Science, 2016, 7: 265 − 281. [16] 权刚. 土壤中的土霉素对蔗糖酶活性的影响[J]. 陕西农业科学, 2019, 65(1): 59 − 61. doi: 10.3969/j.issn.0488-5368.2019.01.014 [17] 石春芳, 王志勇, 冷小云, 等. 土壤磷酸酶活性测定方法的改进[J]. 实验技术与管理, 2016, 33(7): 48 − 49. [18] 赵静, 韩甜甜, 谢兴斌, 等. 酸化梨园土壤酶活性与土壤理化性质之间的关系[J]. 水土保持学报, 2011, 25(4): 115 − 120. [19] 刘淑英. 有机无机肥配施对灌耕灰钙土碱性磷酸酶和土壤磷素的影响[J]. 土壤通报, 2011(3): 670 − 675. [20] 耿玉清, 白翠霞, 赵广亮, 等. 土壤磷酸酶活性及其与有机磷组分的相关性[J]. 北京林业大学学报, 2008(Z2): 139 − 143. [21] 叶飞, 牛高华, 刘惠芬, 等. 转基因棉花种植对根际土壤酶活性的影响[J]. 华北农学报, 2008, 23(4): 201 − 203. doi: 10.7668/hbnxb.2008.04.045 [22] 刘岩. 转基因亚麻根系分泌物对土壤酶活性的影响[J]. 安徽农业科学, 2015, 43(35): 264 − 266. doi: 10.3969/j.issn.0517-6611.2015.35.099 [23] 阮妙鸿, 许燕, 郑瑶, 等. 转ScMV-CP基因甘蔗对根际土壤酶活性及微生物的影响[J]. 中国农学通报, 2007, 23(4): 381 − 386. doi: 10.3969/j.issn.1000-6850.2007.04.088 -




 下载:
下载: